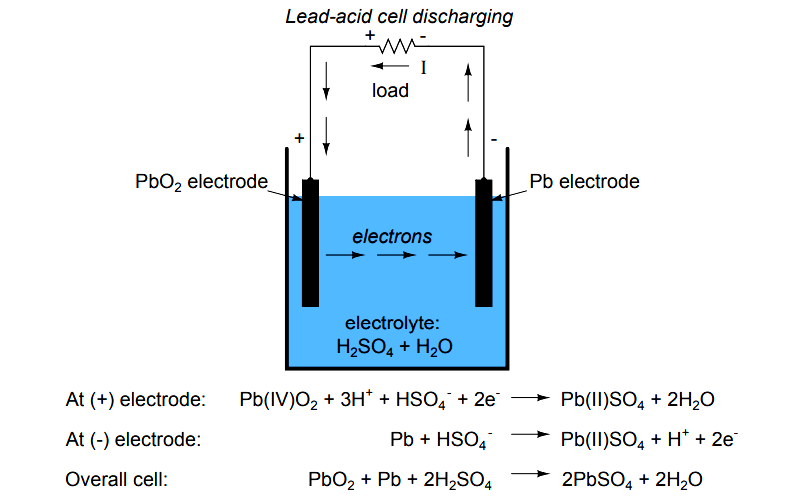
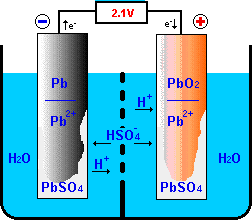
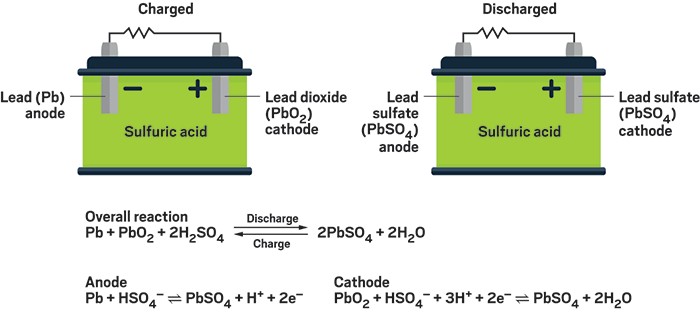
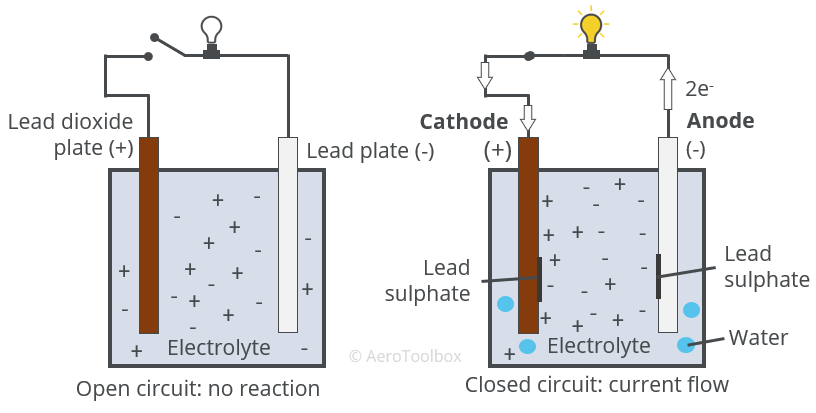
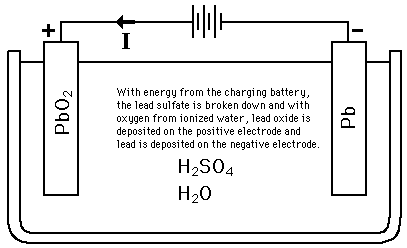
Question Video: Identifying the Reaction That Occurs at the Positive Plate of a Lead-Acid Accumulator Battery During the Discharge Phase | Nagwa

Question Video: Identifying the Overall Equation for the Reaction That Occurs When Lead–Acid Batteries Discharge | Nagwa
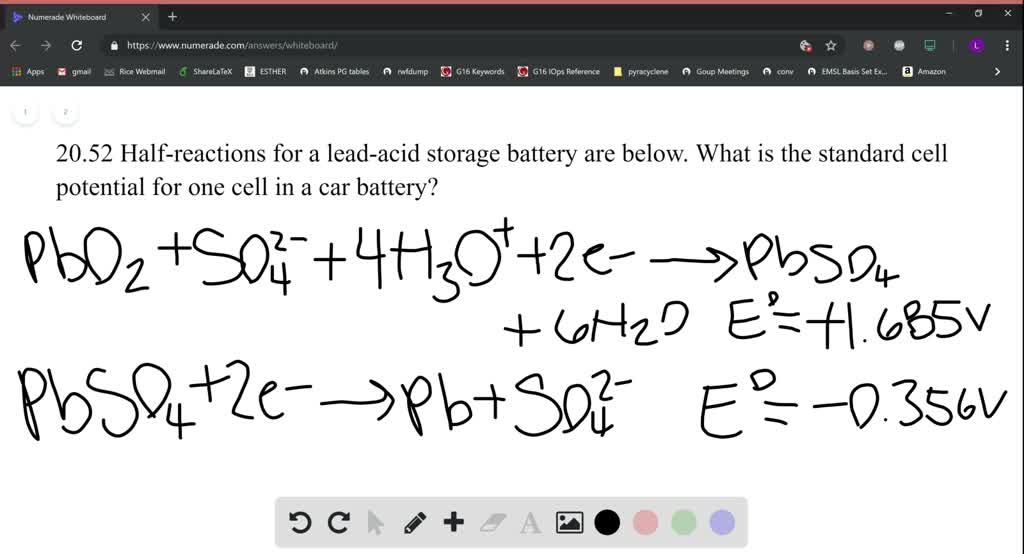
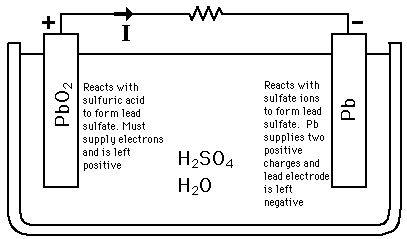
SOLVED:Half-reactions for a lead-acid storage battery are below. PbO2(s)+SO4^2-(aq)+4 H3O^+(aq)+2 e^-→ PbSO4(s)+6 H2O(l) E^0=+1.685 V PbSO4(s)+2 e^-→Pb(s)+SO4^2-(aq) E^0=-0.356 V What is the standard cell potential for one cell in a car battery?